- CasNo98-86-2
- Molecular FormulaC8H8O
- Molecular Weight120.151
- Purity
- Appearanceclear to light yellow liquid
- Packing
- Apply

Your Location:Home > Products > Organic Chemicals



|
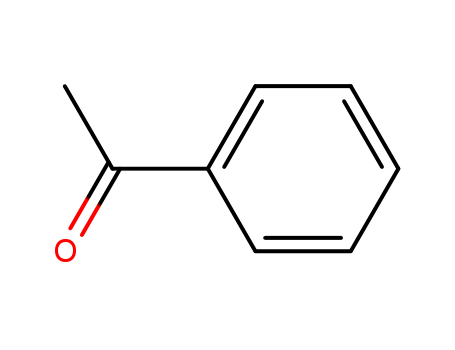
Chemical structure |
Acetophenone is an aromatic ketone with a phenyl ring attached to a carbonyl group. |
|
Production Methods |
Acetophenone can be synthesized industrially through various methods, including the oxidation of ethylbenzene and isopropylbenzene. The use of heterogeneous Ti–Zr–Co alloy catalyst for the oxidation of ethylbenzene to acetophenone is a recent development.[2] |
|
Safety Profile |
Poison by intraperitoneal andsubcutaneous routes. Moderately toxic by ingestion. A skinand severe eye irritant. Mutation data reported. Narcotic inhigh concentration. A hypnotic. Flammable liquid. To fightfire, use foam, CO2, dry chemical. When heated |
InChI:InChI=1/C8H8O/c1-7(9)8-5-3-2-4-6-8/h2-6H,1H3
A new efficient heterogeneous catalyst i...
Sodium percarbonate has been found to be...
An effective method for the debrominatio...
-
The development of a highly efficient st...
-
A new concept for selectivity control in...
The gas phase basicities of m,p-substitu...
A kinetic investigation into the origin ...
Heterogenized photoredox catalysts provi...
-
Coordination and steric environment arou...
A study was conducted to demonstrate the...
The mechanism for the reduction of the α...
-
A mononuclear manganese(iii)-peroxo comp...
Iridium-catalyzed boron-hydrogen bond in...
N-Graphitic-Modified cobalt nanoparticle...
Synthesis of size-controlled Au nanopart...
Bromomagnesium diphenylcuprate and iodom...
An efficient method for the 1,6-conjugat...
-
A new highly selective and environment-f...
-
(Chemical Equation Presented) Fast and f...
A series of α-halocarbonyl compounds are...
The silver-catalyzed reaction of silyl e...
Earlier work revealed that metal-superox...
Several aspects of the mechanism of the ...
The influence of β-substituents on closu...
Tosylhydrazones of carbonyl compounds we...
The formation and detailed spectroscopic...
Recently, we have reported several catal...
Mononuclear MnIII-peroxo and dinuclear b...
(R)-1-phenylethanol is an important subs...
The toxic Hg(II)being released excessive...
An NH4I-based reductive system has been ...
The ruthenium-catalyzed flash oxidation ...
The use of a combined Lewis acid/base sy...
Manganese(III)-peroxo complexes are invo...
5-Hydroxymethylfurfural oxidase (HMFO) i...
Treatment of tosylhydrazones of ketones ...
A series of sulfonated carbon materials ...
Three arene-ruthenium(II) complexes bear...
Cumyl hydroperoxide (CHP) is an importan...
Synthesis of covalent organic frameworks...
In the present work, a highly efficient ...
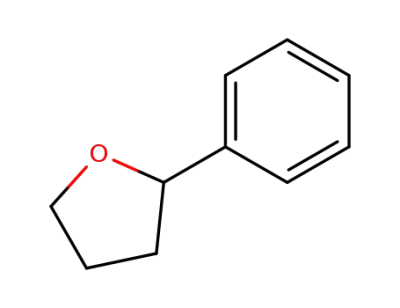
tetrahydro-2-phenylfuran

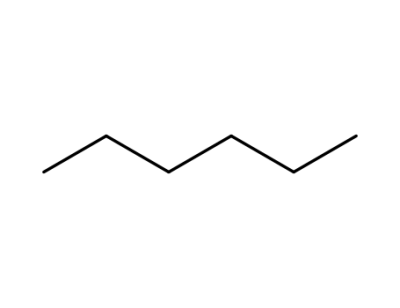
hexane

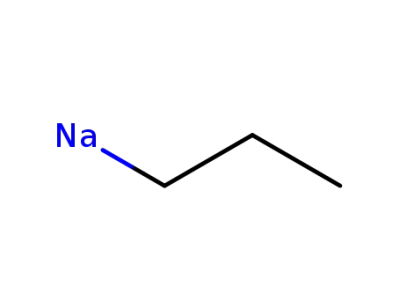
propyl sodium

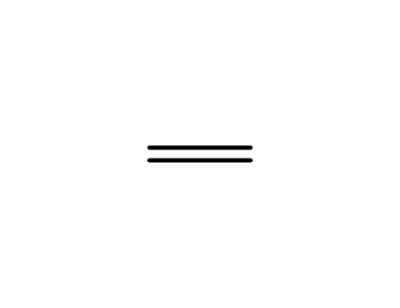
ethene

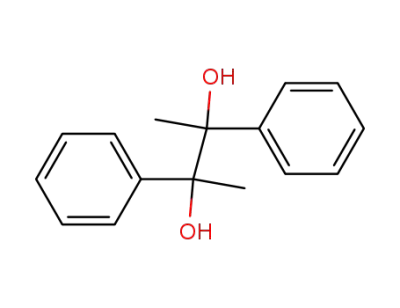
2,3-diphenyl-2,3-butanediol

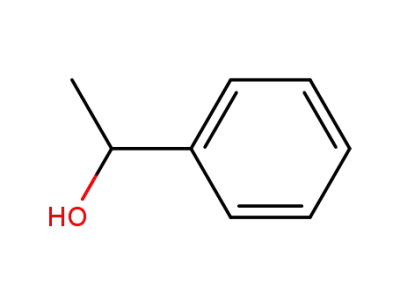
1-Phenylethanol

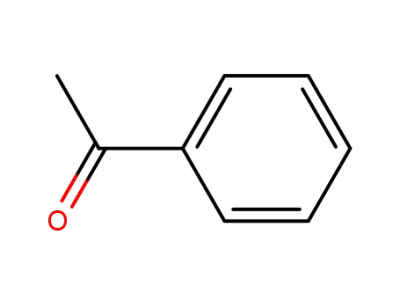
acetophenone
| Conditions | Yield |
|---|---|
|
weiteres Produkt: Propan;
|

2,3-diphenyl-2,3-butanediol


1-Phenylethanol


acetophenone
| Conditions | Yield |
|---|---|
|
at 280 - 300 ℃;
im geschlossenen Rohr;
|

diazomethane
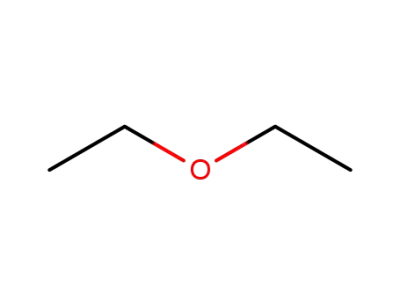
diethyl ether
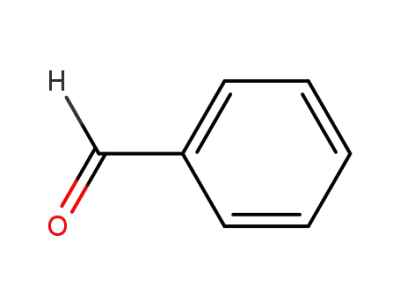
benzaldehyde
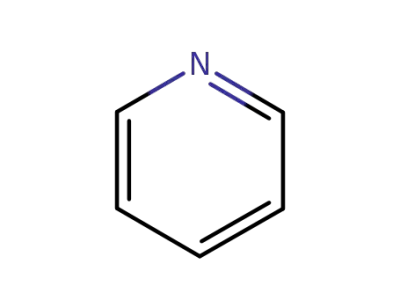
pyridine
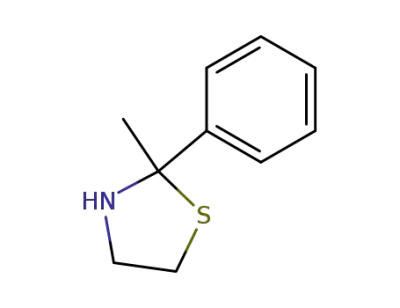
2-methyl-2-phenylthiazolidine
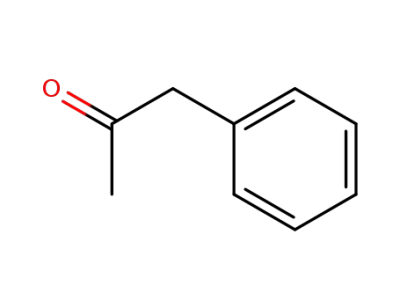
1-phenyl-acetone
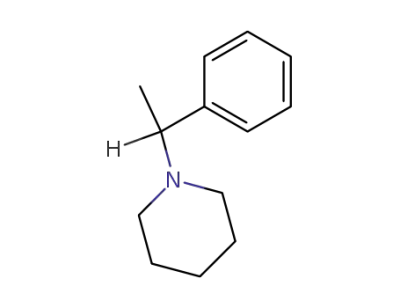
1-(1-phenylethyl)piperidine
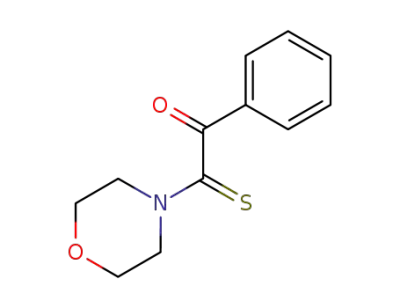
2-morpholino-1-phenyl-2-thioxoethanone
